


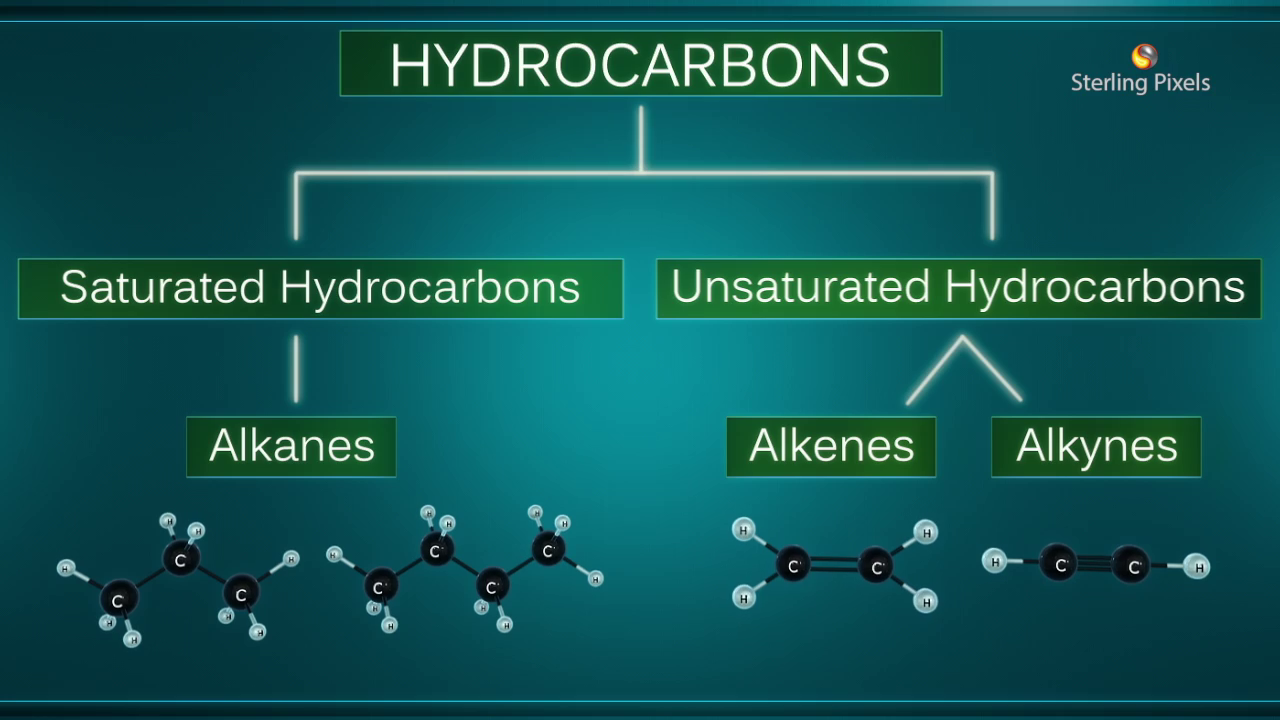
Hydrocarbons are compounds formed from carbon and hydrogen.
They are of two types, namely, saturated hydrocarbons and unsaturated hydrocarbons.
Saturated hydrocarbons, like alkanes, have a single bond between any two neighbouring carbon atoms, for example, propane and butane.
Unsaturated hydrocarbons, like alkenes and alkynes, have double and triple bonds between neighbouring carbon atoms, for example, ethene and ethyne.
Let’s study about the combustion reaction of hydrocarbons.
Combustion reaction is the process of burning of a carbon compound in air to give carbon dioxide, water, heat, and light.
For example, when wood burns in air, the carbon present in the wood reacts with the oxygen present in the air, releasing carbon dioxide and water, along with heat and light.

When saturated hydrocarbons burn, they produce a blue, non-sooty flame.
Let’s study this with the help of an example.
Liquefied petroleum gas, also known as LPG, is a fuel used for cooking.
LPG mainly contains butane, along with small amounts of propane.
Let’s observe the Bunsen burner.
It has air holes near its base, which can be opened or closed with the help of a sliding valve.
Let us first keep the air holes fully open and light the burner.
On burning LPG in air, the propane and butane in it react with the molecular oxygen to form carbon dioxide and water vapour, besides releasing heat and light.

We see that the gas burns with a blue, non-sooty flame and does not give out black smoke.
This is because the percentage of carbon in butane, which is comparably low, is completely oxidized by the oxygen present in the air.
As a result, the bottom of the cooking utensils remains clean from the outside.
Now, reduce the size of the air holes bit-by-bit, thus reducing the amount of air entering the burner.
Due to this, the gas in the burner burns with a sooty flame.

Now close the air holes completely.
The gas burns with a highly-sooty flame, producing thick, black smoke.
This happens because of incomplete combustion of the saturated hydrocarbon in the air.

As a result, the bottom of the cooking utensils gets blackened from the outside.
Unsaturated hydrocarbons like ethyne, also known as acetylene, burn to produce a yellow, sooty flame due to incomplete combustion in air.
The flame is sooty because the percentage of carbon is comparatively higher than that of alkanes and so does not get completely oxidized in air.
However, when acetylene and pure oxygen burn, a non-sooty, blue flame is produced.

This is also known as oxi-acetylene flame and it is used for cutting and welding metals.